Sampling for PFAS Requires Caution
What makes a PFAS investigation different? The answer is the potential for sample contamination from commonly used consumer products and sampling materials.
By Scott Bell, Senior Environmental Engineer/Vice President (Ann Arbor, MI )
July 10, 2018
Recent concern over per- and polyfluoroalkyl substances (PFAS) in the environment has led to a dramatic increase in the number of ongoing investigations to verify the presence or absence of PFAS, to identify sources, and to determine the extent of environmental impacts. One of the most challenging aspects of investigating PFAS contamination is the very low concentrations that require measurement, usually in the parts per trillion. Granted, other chemicals commonly investigated also require quantification at very low levels. What makes a PFAS investigation different, however, is the potential for accidental contamination of environmental samples during sample collection because of their presence in so many commonly used consumer products and sampling materials. This article discusses some of the potential sources of sample cross-contamination, and shares some findings from LimnoTech projects and the technical literature to help others ensure sample integrity during PFAS investigations.
The U.S. Department of Defense probably has the longest record of experience with PFAS site investigation, largely due to the widespread use of PFAS-containing aqueous film-forming foam (AFFF) for firefighting at Navy, Air Force, Air National Guard, and other military air bases. To address concerns about quality assurance in PFAS investigations, the commander of the Naval Facilities Engineering Command (NAVFAC) issued a memorandum titled “Perflourinated Alkyl Compounds (PFCs) Interim Guidance/Frequently Asked Questions” in January 2015, later updated in September 2017. This lengthy memo covers a number of topics, including a response to the question “Are there special sampling techniques for these chemicals?” The response includes a list of prohibited materials such as Post-it Notes, Tyvek suits, water-resistant clothing, blue ice and Teflon®. Since the publication of the NAVFAC document, many other organizations have published similar and more detailed sampling guidance documents. This body of information has informed many standard operating procedures for PFAS investigation, including LimnoTech’s.
A question that has arisen on some of our projects is “What is the optimal level of clean sampling?” For example, we require our field personnel to avoid use of insect repellent (and other products) during sampling activities, but does it really matter? In a recent research article published online in an issue of Remediation, investigators exposed articles of clothing to commonly used insect repellents, then immersed the clothing in PFAS-free water and tested the water for PFAS to see if the banned insect repellent leached PFAS to the water, simulating sample contamination. In that case, all water samples tested non-detect (<2.5 ng/l) for all PFAS analyzed. In another case, a well-known manufacturer of environmental investigation equipment tested a popular submersible groundwater sampling pump and various tubing to determine whether they contributed PFAS to water during sampling. The pump includes a bladder made of low-density polyethylene (LDPE), a material commonly found on PFAS sampling material exclusion lists. That test showed no detectable PFAS in the water (<1 ng/l for most compounds) after exposure to the sampling materials.
A question that has arisen on some of our projects is “What is the optimal level of clean sampling?” For example, we require our sampling personnel to avoid use of insect repellent (and other products) during sampling activities, but does it really matter?
The inclusion of Teflon on the list of prohibited PFAS sampling materials is particularly noteworthy because Teflon® is used in many sampling products including tubing, pumps, and direct-push soil samplers. Teflon® is a brand name for polytetrafluoroethylene (PTFE), a chemical that has been used to create nonstick surfaces for many decades. The current concern associated with Teflon® is that perfluorooctanoic acid (PFOA), a regulated PFAS, is used in the manufacture of Teflon®, and the fear is Teflon® materials used in sampling can leach PFOA or other PFAS into the sample being collected. For two recent projects, LimnoTech planned to use a direct-push sampling method, but the sample liners used by most direct-push samplers are made of Teflon®. To test the potential for these liners to contaminate our samples, we collected equipment rinse blanks on the liners by exposing PFAS-free water (certified by the testing laboratory to be PFAS-free) to the Teflon® samplers. In both cases, these equipment blanks came back with two types of PFAS (PFBA and PFHxS) detected between the method detection limit and the reporting limit, indicating very low-level PFAS contamination. The consistency of these results for blanks collected on two separate occasions at different sites with different batches of PFAS-free water is notable. Although the estimated concentrations in the blanks were below the reporting limits, the results suggest that PFAS can leach into water samples. Further tests are planned with longer durations of exposure to evaluate the effects of contact time on blank concentrations.
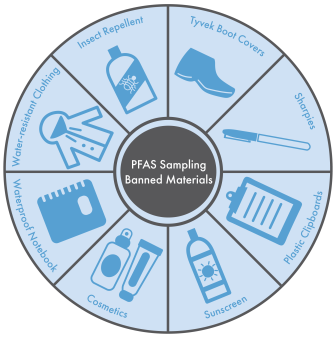
Banned materials for PFAS investigations include sunscreen, insect repellent, Sharpie markers, latex gloves, water-resistant clothing, blue ice, cosmetics, etc. Disclaimer: The items depicted in the image are intended to provide a visual example of banned materials. The inclusion of a specific product does not indicate whether PFAS is in the actual product.
Another potential source of sample contamination is public water (tap water). During site investigations, it is not uncommon to use public water supplies for equipment decontamination between samples, as well as in some drilling processes. The substantial quantities of water required for these activities make it impractical to use laboratory-supplied PFAS-free water, so we must rely on available public water supplies. However, we know it is not unusual to find PFAS in public water supplies, albeit usually at low levels. In Michigan, for example, a number of public water supplies have already been found to contain levels of PFAS below Federal lifetime health advisory levels. For a recent PFAS investigation, we needed to decontaminate drilling equipment between borings, and the only practical way to do so was to use the local water supply. We tested the water ahead of time for certain types of PFAS. When we analyzed the water supply, we found perfluorooctane sulfonate (PFOS) at concentrations slightly above the practical quantitation limit (PQL) of 1.9 ng/l in both the original sample and a blind duplicate.
Another potential source of sample contamination is public water (tap water)… When we analyzed the water supply, we found perfluorooctane sulfonate (PFOS) at concentrations slightly above the practical quantitation limit (PQL) of 1.9 ng/l in both the original sample and a blind duplicate.
So where does all this leave us? Is it really necessary to exercise extreme caution during PFAS investigations? Sometimes the question is phrased in the context of the cost for collecting additional quality assurance samples (i.e., equipment blanks, field blanks, etc.). With analytical costs in the hundreds of dollars per sample, the question is understandable. There is certainly evidence that PFAS can exist at low concentrations in sampling materials and consumer products. There is also evidence that PFAS can leach from these materials, potentially resulting in “false positives.” Likewise, the presence of low concentrations of PFAS in water supplies is a concern when that water is used during PFAS investigations. EPA has expressly prohibited blank subtraction as a contamination correction in Analytical Method 537 (US EPA, 2009). When researching the technical literature for this article, we were unable to find examples of sample contamination from sampling materials or consumer products used during sampling that caused contamination of samples at concentrations above levels of regulatory concern, but that doesn’t mean examples are not out there and have yet to be published. Our recommendation is to proceed with caution. Field personnel need to avoid the use of sampling materials that may contain PFAS and make sure to collect appropriate quality assurance samples as well. The old phrase “better safe than sorry” is applicable here. The cost of taking appropriate precautions during sampling and collecting adequate and appropriate quality assurance samples to quantify potential contamination sources and ensure defensible data will usually be far less than the cost of having to redo some or all of a PFAS investigation.
This article is the second in a series of articles authored by LimnoTech staff on PFAS-related issues. Follow us on LinkedIn or Twitter (@LimnoTech) and check the News and Media page on our website for more information and updates.
If you want to discuss your PFAS-related problem, please feel free to contact me at sbell@limno.com.
Scott Bell , PE, BCEE, is a senior environmental engineer and Vice President at LimnoTech. Scott has extensive experience in water resources engineering analysis and has been with LimnoTech since 1992. In addition to managing staff, projects, and clients, Scott has technical expertise in environmental remediation and restoration, hydrologic and hydraulic engineering, wastewater discharge impact analysis and mitigation, and stormwater management. He is currently supporting industrial and civilian aviation clients in Michigan with their PFAS-related problems.